So, first, they can be difficult to observe and measure. Intermolecular forces are often very weak and exist within a microscopic molecule. For instance, students often mix the two terms intermolecular and intermolecular forces. The types of intermolecular forces are often confusing to students because so many identical terms are used to describe them. 3 Reasons Why Study of Intermolecular forces can be tricky for studentsįirst, let’s explore the three reasons why students find the concept of intermolecular tricky. So, read on to learn five ways through which you can make intermolecular forces an interesting topic to teach. As a teacher, you may find it hard to explain it to students. Intermolecular force is one of the fundamental concepts that you will have to teach in an introductory chemistry course. Image from Labster's lab simulation on Intermolecular Forces. It falls under the category of permanent dipole-dipole forces as it is an attractive force between permanent dipoles. 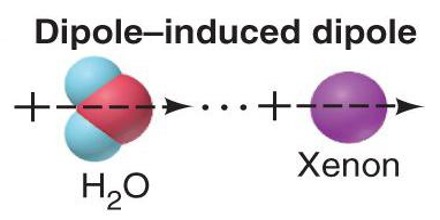
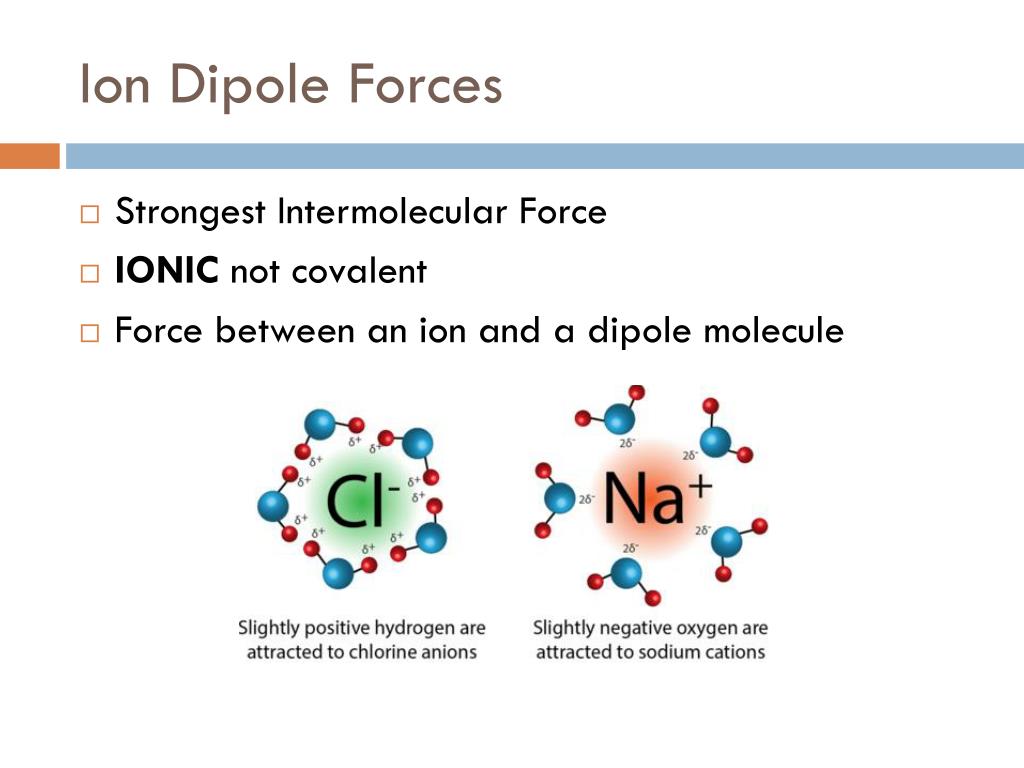
Hydrogen bonding is the most powerful intermolecular force. Second, this dipole must approach another atom of oxygen, fluorine, or nitrogen to form a hydrogen bond. First, it must form a strong molecular dipole by a covalent bond with either oxygen, fluorine, or nitrogen. However, hydrogen alone is insufficient for hydrogen bonding. Hydrogen Bonding, as its name suggests, is a bond between hydrogen molecules. The force of attraction between these molecules is termed dipole-dipole interaction. The dipole can be permanent or induced due to the presence of another ionic molecule. This polarized molecule is called a dipole. In polar molecules, one end is slightly negatively charged and the other is slightly positively charged due to the electronegativity difference between the two bonding atoms. These forces exist in almost all molecules to some extent.ĭipole-Dipole forces are a property of polar molecules. Van der Waals forces are weak intermolecular forces present in between neutral or nonpolar molecules. Intermolecular forces are categorized into three major types, Van der Waal forces, dipole-dipole forces, and hydrogen bonding. The stronger the intermolecular forces, the higher the melting and boiling points are. These forces are important because they help chemists determine the physical properties of a substance, such as its state, its melting and boiling point, etc. 
Intermolecular forces are the forces of attraction between molecules.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
